How We Develop & Maintain PQA Measures
PQA uses a systematic, transparent and consensus-based process to conceptualize, specify, test, endorse and maintain measures of medication use quality in the domains of Appropriate Use, Safety, Adherence and Medication Management Services.
The PQA Measure Life Cycle aligns with the CMS Measures Management System: Blueprint Measure Lifecycle, the “gold standard” process for measure development and maintenance. The life cycle consists of five phases and balances rigor with flexibility. It centers on standard criteria for evaluating measures, which include importance, scientific acceptability, feasibility and usability—ensuring that PQA measures are evidence-based, precisely specified, valid, reliable, feasible and usable while advancing national quality goals.
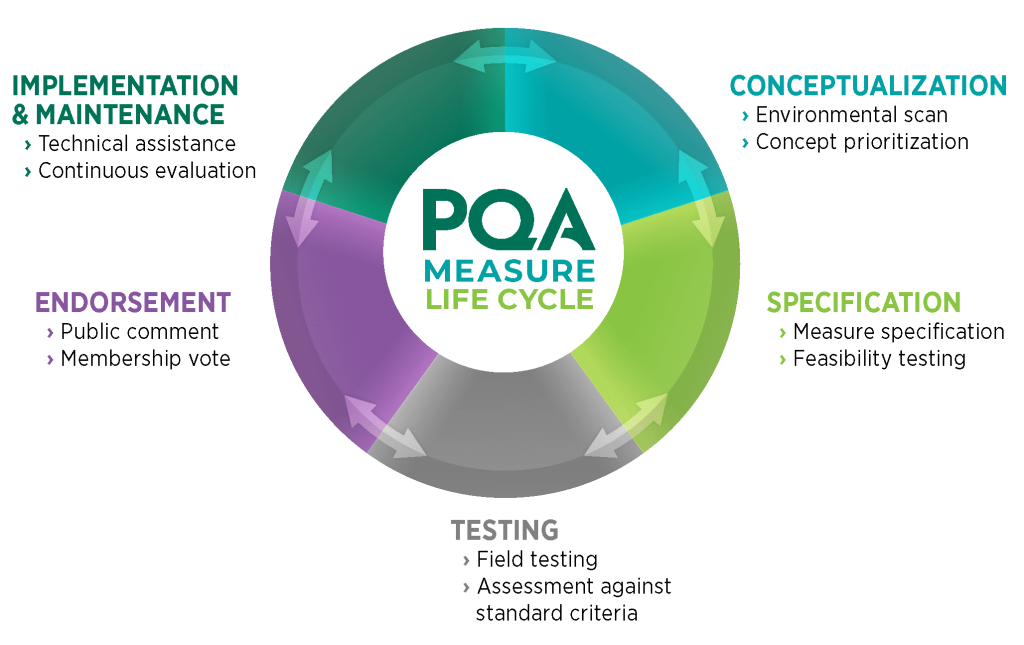
Conceptualization
Specification
Testing
Endorsement
Implementation & Maintenance
Measure conceptualization involves information gathering and business case development to identify and prioritize measures for development. This focuses resources on high-impact measures that address a quality measurement gap.
Measure specification entails the creation and refinement of detailed specifications for how to calculate the measure. This stage may involve initial feasibility testing (sometimes referred to as alpha testing).
Field testing involves applying measure specifications to test data representative of the intended measure population to evaluate the measure’s scientific acceptability and gauge opportunity for improvement.
After testing, measures are assessed by an expert panel against standard measure evaluation criteria, and if appropriate, advance to public comment and a PQA membership vote.
After endorsement, PQA supports implementation with outreach, education, and technical assistance. PQA continually evaluates measures to ensure they stay current and impactful as evidence evolves.
Standard Measure Criteria
Importance
The measure addresses a priority area, is evidence-based, and has the potential to improve health care quality and outcomes.
Scientific Acceptability
The measure, when implemented, will yield consistent (reliable) and credible (valid) results for the intended area of measurement.
Feasibility
The data required to calculate the measure are readily available, accurate and retrievable without undue burden.
Usability
There is opportunity for the measure to be implemented with results supporting both accountability and performance improvement.
Involvement in PQA’s Measure Life Cycle
Developing consensus-based measures requires the right perspectives and expertise. PQA’s multistakeholder membership informs all phases of the life cycle through quarterly membership meetings, nomination based technical panels and advisory groups, measure testing, and measure endorsement and retirement voting. PQA seeks feedback from both members and non-members for additional transparency during the life cycle through public comment periods.
PQA engages patients, caregivers and patient advocates throughout the life cycle because their perspectives, insights, and feedback are essential for meaningful, person-centered quality measures. Patient partners are included in all PQA nomination-based panels and groups.
Engagement Opportunities
Measure Concept Advisory Groups (MCAGs) are nomination-based and convened as needed to advise PQA during measure conceptualization to support prioritization for development.
Technical Expert Panels (TEPs) are nomination-based and convened as needed for subject matter experts to address key questions identified during measure specification and feasibility testing.
The Quality Metrics Expert Panel (QMEP) is a standing, nomination-based panel of subject matter experts that reviews measure specifications and updates, measure testing plans and testing results, and assesses measures for endorsement and retirement consideration by PQA membership.
The Measure Update Panel (MUP) is a standing, nomination-based panel of subject matter experts that reviews PQA-endorsed measures to ensure they continue to align with current clinical evidence and guidelines and that the measures remain viable for use.
Field Testing (sometimes referred to as beta testing) is a collaborative process between PQA and member organizations that volunteer data and analytic resources to validate clarity, reproducibility, and feasibility of draft or updated specifications.
Public Comment Periods are conducted to support transparency and broad stakeholder input throughout the measure life cycle. Open to members and the public, PQA solicits feedback on measures considered for development, endorsement or retirement, and substantive updates made to measures in use.
PQA Member Webinars, held quarterly and open to all PQA members, provide the opportunity for PQA staff to communicate updates on PQA’s measurement and research activities and receive broad stakeholder feedback from members.
PQA member organizations have the opportunity to review measure materials and vote on measures under consideration for endorsement or retirement. Each organization may cast one vote per measure, indicating support, opposition, or abstention.

